Beta Thickness Gauging
An ATI gauge uses the attenuation of beta particles to measure the thickness or weight of various materials, such as plastics, paper, and metal. ATI’s gauges can be used for measuring coat weight, basis weight , thickness, and detection. ATI’s gauges can provide measurements from a basis weight range of 1 gsm to 8,500 gsm with the fastest response time.
What components make up a beta gauge?
A beta gauge consists of two basic components - a source of radiation, and a radiation detector. The web to be measured is placed between the source and detector. In addition, some sort of computer is used to process the information from the detector, and convert it into a measurement. The illustration below shows the basic setup:
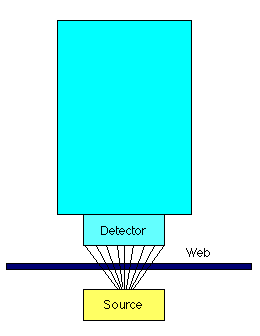
What are beta particles?
Beta particles are nothing more then fast moving electrons, emitted from certain radioactive isotopes, called sources. These electrons are emitted when the atom undergoes a decay.
Are they dangerous?
Like all things in life, they can be dangerous in excess. Compared to gamma rays, beta particles are much safer because they do not travel as far, and they are easily stopped by several feet of air, or thin plastic. This makes it quite easy to install a shield around the gauge, if personnel are working nearby.
In addition, ATI uses very small activity (size) sources. While conventional beta gauges use sources with hundreds or thousands of milli-curies of activity, ATI sources typically range from 0.5 to 3 milli-curies. This substantially reduces the amount of radiation present around the gauge.
How are beta particles used to measure material?
When they strike material, some of them will pass through, while others will be stopped. The thicker (or more dense) the material, the greater the chance a particle will be stopped. By measuring the ratio of the number of particles that pass through the material to the number without any material, the thickness (or weight) of the material can be determined.
How do conventional beta gauges work?
Conventional beta gauges use an ionization chamber to detect the beta particles. This is essentially a can, filled with an inert gas such as argon. The body of the can is connected to a high voltage (around 500V), and an electrode in the can is connected to a sensitive amplifier. Beta particles entering the can ionize gas atoms, the freed electrons are attracted to the electrode, and the extremely weak current (typically several nanoamperes at most) is amplified, and converted to a voltage, proportional to the number of beta particles entering the detector. The voltage is an analog signal and does not provide a real time measurement. The measurement contains a percentage of the previous measurement.
What's different about ATI's technique?
ATI's patented technique uses a large surface area PIN diode to directly convert each beta particle into a pulse of current, which is then counted. Since each beta particle is individually detected and counted, the efficiency is near 100%. This allows ATI to use much smaller radioactive sources (typically 0.5 to 3 millicuries, vs 300 to 1000 millicuries used by gauges employing ionization chambers). By counting pulses, ATI provides a real time digital measurement. Each measurement is independent from the previous measurement providing a 100% step response.
Why do beta gauges have noise in their measurement?
This is due to the fact that the decay of an atom is a random event. If we have say 1000 atoms, each with a half life of 1 year, then over the course of a year, we would expect half of them (500) to decay. But not exactly 500 of them will decay. It gets a little technical, but bear with us...
The standard deviation of the number which will decay is equal to the square root of the expected number. The square root of 500 is about 22. That means that, 67% of the time, we would expect 500 +/- 22 atoms to decay, or anywhere from 478 to 522 atoms. So, if we place a material in the beam that only stops say 10% of the beta particles, then it stops 50 of them, and we would expect to count 450 particles, with a standard deviation of 21. But it's clear that there really might be 429 to 471 particles, one standard deviation is 4.8%. The randomness of the number of beta particles detected is large compared to the change caused by the material. So we cannot make a good measurement.
By increasing the number of atoms decaying, we can improve the measurement. If we instead have 500,000 atoms decaying, then a material that stops 10% of the beta particles would stop 50,000 of them, and there would be 450,000 to detect. The standard deviation is 671, or 0.15%, much less than the previous example.
This lets us make a better measurement. So, by increasing the size of the source (or using a better detector that counts more of the incoming beta particles) we can improve the precision of the measurement. We can also do this by averaging the measurement over a longer time period. This gives us more particles to count, so we get a better average, with less variation. We can also reduce the air gap (distance between the source and detector). Finally, and perhaps most important, we can use the appropriate source for the measuring range.
What thickness or basis weight range can be measured?
In order to make an accurate measurement, it is important that the material not be so heavy that it stops all (or too many) of the beta particles. It is also important that it stops enough of them. If it is too light in weight, so few of the beta particles will be stopped that it will be difficult to measure the number that are stopped. In other words, only a very small number of beat particles will be stopped.
The chance that a beta particle will make it through the material depends on how heavy the material is, and on the speed of the beta particle. A faster moving particle has a better chance of pasing through the material. Different sources produce beta particles with different speeds. So, by selecting the source that produces beta particles of the correct speed, we can match that speed to the weight of the material we are trying to measure. This is why several different sources are used in gauging.
There are three commonly used beta sources:
Promethium (Pm147)
This is the lowest energy beta source commonly used, it is suitable for measurements up to around 275 GSM.
Krypton (Kr85)
This is a medium energy beat source. It is suitable for measurements in the range of 150 to 1500 GSM.
Strontium (Sr90)
This is the highest energy beta source commonly used. It is suitable for measurements in the range of 1000 to 8000 GSM.
The above ranges are in GSM, grams per square meter. To convert from GSM to thickness in microns, multiply by the density of the material. To convert from microns to mils (thousanths of an inch) divide by 25.4.
For most plastics or other materials with a density of one, one mil (thousandth of an inch) is 25.4 GSM.
